Fundamental Particles need to get together to form matter. How do they do that?
Quarks combine to make protons and neutrons.

The protons are both positive and they repel each other. The strong nuclear force holds the nucleus together.

Nucleus is positive - The electrons are negative. Together they make up the atom.
Atoms like to have equal numbers of positive protons and negative electrons.
If you need to practice putting protons and nuetrons together, try this simulation.
Click the Image or HERE to play.
The positive nucleus and the negative electrons have an electrostatic Force between them. It is known as Coulomb;s Law. The equation is below.
The bigger the charge, the greater the force,
The greater the distance between the charges the, smaller the force. Does that make sense mathematically to to you?

Electrons are moving- They have energy.
From looking at spectrum and experiments, we know that the electrons can be in different energy levels.
It is easiest to look at a Hydrogen atom. It has a proton in the nucleus and a single electron. When the electron is in the 1st energy level, we say it is at ground state.
We can calculate the energy by using the equation E=13.6eV (1/n^2)
eV are electron volts - they are a unit of energy.
n is the energy level
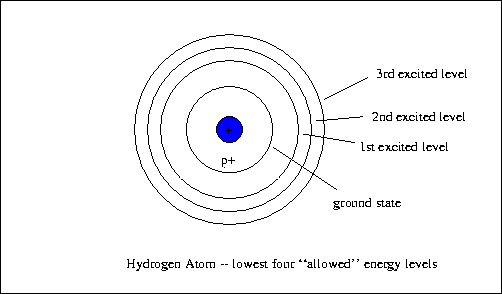
How does an electron get to a higher level?
You add energy- When we looked at the spectra, we plugged the tube in and electricity produced teh photons that moved the electron to a higher level. When the electron is in a higher energ level, it is an excited atom.


Another reading and video - The Bohr Model and Atomic Spectra with a Quiz
This video explains the content of this packet but also gets into some of the information in the next Sophia tutorial